32nrt

First Law Of Thermodynamics Internal Energy Video Khan Academy
2

U 3 2 Nrt Proof Ke 3 2nrt Ke Equals 3 Divide By 2 Nrt For A Monoatomic Ideal Gas Kisembo Youtube
2
2

U 3 2 Nrt Proof Ke 3 2nrt Ke Equals 3 Divide By 2 Nrt For A Monoatomic Ideal Gas Kisembo Youtube
• What is the molar volume of an ideal gas at STP?.
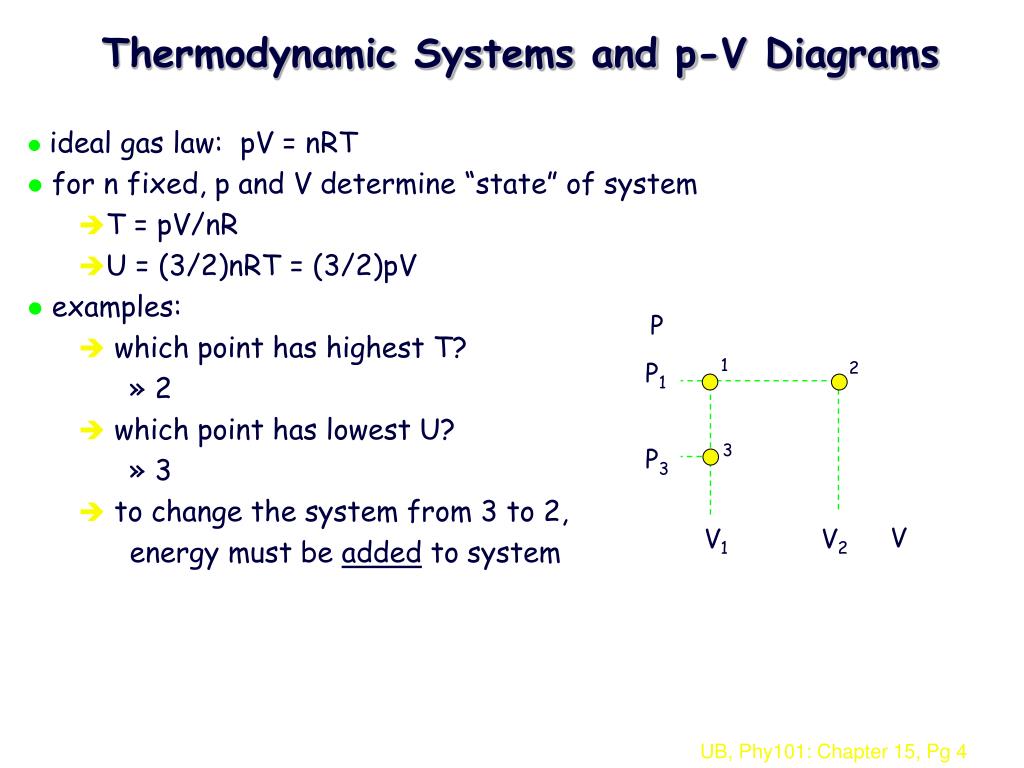
32nrt. Ok Pressure * Volume = Numbe rof moles * molar gas constant * Temperature I have got it down to 1/2m*meansquaredspeed = (3/2nRT)/Number of particles I know i am suppose to use avogadro equation and then use the Boltzman constant but the numbers are not popping out Can someone just do the maths for me please to get 1/2m*meanssquaredspeed = 3/2kT Thank you!. And a whole lot more!. Free math problem solver answers your algebra, geometry, trigonometry, calculus, and statistics homework questions with stepbystep explanations, just like a math tutor.
Are your accessibility or localization needs BIG?. Chemical reactions and stoichiometry;. 6 Work done on or by a gas Imagine compressing a gas by exerting a.
Ek=3/2PV=3/2nRT=3/2NkBT Given the COVID19 pandemic, call ahead to verify hours, and remember to practice social distancing No tips and reviews Log in to leave a tip here Post No tips yet Write a short note about what you liked, what to order, or other helpful advice for visitors. Substituting back R/N A instead of k B gives E Ktotal = 3/2 x N/N A x R x T = 3/2nRT = 3/2pV (since pV=nRT) So E Ktotal = 3/2nRT and E Ktotal = 3/2pV Documents 32 Modelling a Gas Useful Links Going into thermodynamics which is in one of the options You don't need to watch this video but it provides a nice link to other ideas. Hi i am working on trying to derive KE (or Etrans)=3/2nRT so i know these steps that ___________ pV= nRT and pV= 1/3 NmV2 and combining them, we get nRT= 1/3Nmv2 also, we know that for translational motion, KE= 1/2Nmv2 ______ i don't know how 2/3 (1 /2 Nmv2) = nRT where did that 2/3 come from??!!.
(3/2)nRT is the translational kinetic energy, and since almost all atoms are in the ground electronic state at low temperature, it is a good expression for internal energy as long as the temperature is low enough that essentially all atoms are in the electronic ground state. U = 3/2nRT It can be derived that the molar specific heat at constant pressure is C p = C v R = 5/2R = 8 J/mol K This C p is greater than the molar specific heat at constant volume C v, because energy must now be supplied not only to raise the temperature of the gas but also for the gas to do work because in this case volume changes. Learn what the first law of thermodynamics is and how to use it.
(How much volume does 1 mole of gas occupy?) nRT (1000 mol)(0006 L atm/mol K)(K). Our intuitive subtitle editor is free!. The trip begins in early morning at a temperature of 15 degrees C By midafternoon, the temperature has increased to 30 degrees C Assuming the pressure remains constant at 100atm, for each mole of helium, calculate (a) The initial and final volumes (b) The change in internal energy, ΔE Hint Helium behaves like an ideal gas, so E=3/2nRT.
Ok Pressure * Volume = Numbe rof moles * molar gas constant * Temperature I have got it down to 1/2m*meansquaredspeed = (3/2nRT)/Number of particles I know i am suppose to use avogadro equation and then use the Boltzman constant but the numbers are not popping out Can someone just do the maths for me please to get 1/2m*meanssquaredspeed = 3/2kT Thank you!. U = 3/2nRT It can be derived that the molar specific heat at constant pressure is C p = C v R = 5/2R = 8 J/mol K This C p is greater than the molar specific heat at constant volume C v, because energy must now be supplied not only to raise the temperature of the gas but also for the gas to do work because in this case volume changes. Substituting back R/N A instead of k B gives E Ktotal = 3/2 x N/N A x R x T = 3/2nRT = 3/2pV (since pV=nRT) So E Ktotal = 3/2nRT and E Ktotal = 3/2pV Documents 32 Modelling a Gas Useful Links Going into thermodynamics which is in one of the options You don't need to watch this video but it provides a nice link to other ideas.
The equipartition principle gives 3/2nRT for the internal energy of a perfect monatomic gas What are the values of {eq}(dU/dV)_{T} {/eq} and {eq}(dH/dV)_{T} {/eq} for a perfect gas?. Amara’s platform and services can help. The change in the internal energy of the gas {eq}\Delta U=386\ \rm J {/eq} The internal energy of a monoatomic ideal gas {eq}U=3/2nRT {/eq}.
Key Takeaways Key Points The ideal gas law assumes that gases are composed of point masses that interact via completely elastic collisions Real gases are made up of particles that occupy a nonzero volume known as the excluded volume. We known U=3/2nRT (monatomic ideal gas), just depends on temperature Most texts assert connecting U and Q with constant volume condition and say"\\Delta U = nCv\\Delta T for any process because of internal energy only depends on temp" I think that statement is very strange Deriving. Ek = 3/2nRT, where R is the molecular gas constant for ideal gases For average kinetic energy of particles, it is Ek = 3/2NkT, where k is the Boltzmannconstant and N the number of gas molecules This leads to Ek = 1/2nMv² = 3/2nRT = 3/2NkT So you get v = (3RT/M)^1/2 by converting the equation 1/2nMv² = 3/2nRT.
Kinetic theory Sections 1311 1315 We're now going to draw on much of what we've learned in this course to understand the motion of gases at the molecular level. Volume of 1 mole of an ideal gas?. The formula U=3/2nRT only works for monotomic substances Every time that more degrees of freedom are added, the 3/2 constant increases So in fact, the potential energy formula should yield correct results had I been using the right one.
Redox reactions and electrochemistry;. • What is the molar volume of an ideal gas at STP?. I had always thought the total kinetic energy in a system is PV = nRT, but today at school I saw someone say it was 3/2 nRT or 3/2 PV Can anyone prove this and explain why PV does not yield the kinetic energy of a system?.
The equipartition principle gives 3/2nRT for the internal energy of a perfect monatomic gas What are the values of {eq}(dU/dV)_{T} {/eq} and {eq}(dH/dV)_{T} {/eq} for a perfect gas?. Of mass reference frame and it is U=3/2nRT=3/2PV • U is called a “state variable” because once the state of the system is specified, such as the number of moles and the temperature or pressure and volume, U has an unique value, that is U is function U(n,T) or U(P,V) The change of a state. I know that subsequently, it will be 2/3KE= nrt and i will get KE=3/2 nRT i just don't KNOW WHERE.
La chaleur spécifique est une propriété liée à l'énergie interne qui est très importante en thermodynamique Les propriétés intensives cv et cp sont définies pour des substances compressibles pures et simples Génie thermique. Demonstração conceitual de que a energia interna de um sistema de gás ideal é de 3/2 PV. An introductory collegelevel chemistry course that explores topics such as atoms, compounds, and ions;.
Volume of 1 mole of an ideal gas?. K tot trans = N 1 2 m ¯ v 2 = 3 2 NkT = 3 2 nRT (347) If the gas only has translational kinetic energy, this is the internal energy of the gas – this is true for ideal monoatomic gases (one atom per molecule) This tells us that the internal energy of an ideal gas depends only on the temperature The root mean square (rms) speed is the square root of the average of the squares of the. Of mass reference frame and it is U=3/2nRT=3/2PV • U is called a “state variable” because once the state of the system is specified, such as the number of moles and the temperature or pressure and volume, U has an unique value, that is U is function U(n,T) or U(P,V) The change of a state.
Le gaz parfait est un modèle thermodynamique décrivant le comportement des gaz réels à basse pression Ce modèle a été développé du milieu du XVII e siècle au milieu du XVIII e siècle et formalisé au XIX e siècle Il est fondé sur l'observation expérimentale selon laquelle tous les gaz tendent vers ce comportement à pression suffisamment basse, quelle que soit la nature. LES GAZ PARFAITS ET REELS 41Le gaz parfait Le gaz parfait est un gaz idéal il correspond à un gaz dilué càd un gaz à pression réduite ;. U 3/2 nR ?.
36k votes, 69 comments 42m members in the dankmemes community dankmemes Press J to jump to the feed Press question mark to learn the rest of the keyboard shortcuts. Correction, il faut utiliser U=3/2nRT et T=mv^2/3k pour un gaz monoatomique, ou U=5/2nRT et T=mv^2/5k pour un gaz diatomique Merci à Duchemin de m'avoir obligé à me relire Reste que dans la modélisation du gaz parfait, on exploite certaines propriétés de la distribution des vitesses de Boltzmann, ce qui risque de conduire à des paradoxes si on les applique à une molécule isolée AC. PV=nRT nRT=(2/3)(N)KE From This we can get KE=3/2nRT/N For unit mole of the gas n=1 and according to BOLTZMAN R/N=K so KE= (3/2) KT Share Cite Improve this answer Follow answered Dec 24 '16 at 600 EngrUzair Hashmi EngrUzair Hashmi 1 $\endgroup$ 1 1.
Le respect de la vie privée de nos utilisateurs étant important pour nous, Lapostenet respecte totalement la confidentialité de vos mails. As KE = 1/2mV^2 so PV=(2/3)(N)KE We know from ideal gas equation ' PV=nRT nRT=(2/3)(N)KE From This we can get KE=3/2nRT/N For unit mole of the gas n=1 and according to BOLTZMAN R/N=K so KE= (3/2) KT. Pay your bill, find free WiFi, check your email, set up your voicemail, program your DVR and more!.
Les cotisations au Club pour 21 sont ouvertes !. Get answers to everything Optimum!. Le montant de la cotisation s’élève à 65 € Adhérez au Club911Net !.
Ok Pressure * Volume = Numbe rof moles * molar gas constant * Temperature I have got it down to 1/2m*meansquaredspeed = (3/2nRT)/Number of particles I know i am suppose to use avogadro equation and then use the Boltzman constant but the numbers are not popping out Can someone just do the maths for me please to get 1/2m*meanssquaredspeed = 3/2kT Thank you!. My thinking was this PAd = some tiny work done by n moles of the gas, d is a tiny distance moved v = Ad = tiny volume expanded or contracted N/n = ratio between total moles. C'est un ensemble de N atomes ou molécules identiques sans interaction entre eux et soumis à une agitation perpétuelle et aléatoire (dite agitation moléculaire ou thermique).
5 Ideal Gas Internal Energy If the room were moving at a high velocity would that mean the internal energy of the gas would be greater?. 3 Adiabatic Process EoS \quad PV^\gamma = const (true only for adiabatic process) Work Done W_{i\rightarrowf. Théorie cinétique des gaz par Gilbert Gastebois Constante de Boltzmann k = 1,380 650 3 × 1023 J/K Constante de Planck h = 6,626 068 74 × 1034 Js 1.
La généalogie de Jésus, par laquelle s’ouvre l’Évangile de Matthieu, énumère quatre femmes dont la présence dans cette succession de générations essentiellement masculines ne peut être expliquée de façon satisfaisante par les seules données bibliques Le recours à la tradition juive permet de comprendre ce que ces femmes pouvaient représenter pour l’auteur du premier. Click sur le bandeau ci dessus ou suivez ce lien pour cotiser. The KE=(1/2)mv2 video link is https//wwwyoutubecom/watch?v=7jqnKbeX5E This video is a very quick, all math, derivation of KE=(3/2)nRT=(3/2)PVThis is.
The ideal gas law, also called the general gas equation, is the equation of state of a hypothetical ideal gasIt is a good approximation of the behavior of many gases under many conditions, although it has several limitations It was first stated by Benoît Paul Émile Clapeyron in 14 as a combination of the empirical Boyle's law, Charles's law, Avogadro's law, and GayLussac's law. 4 Ideal Gas Internal Energy What is the internal energy of 30 moles of oxygen gas at room temperature?. Germany at the Accession of Charles V Charles V (24 February 1500 21 September 1558), reigned over the Holy Roman Empire since 1519 As.
Free math problem solver answers your algebra, geometry, trigonometry, calculus, and statistics homework questions with stepbystep explanations, just like a math tutor. 1 Introduction Apple (Malus domestica) is one of the essential commercial fruit tree cultivated across the world, and China is the leading apple producing countryOver the past few decades, the development of dwarf apple rootstock is a prime pursuit for the modern apple industry However, China apple breeding program has not been established (Li et al, 15). U = 3/2nRT It can be derived that the molar specific heat at constant pressure is C p = C v R = 5/2R = 8 J/mol K This C p is greater than the molar specific heat at constant volume C v, because energy must now be supplied not only to raise the temperature of the gas but also for the gas to do work because in this case volume changes.
This page looks at how and why real gases differ from ideal gases, and takes a brief look at the van der Waals equation If you have come straight to this page via a search engine, it might be a good idea to read the page about ideal gases first Real gases v ideal gases. Want to caption or translate video?. Are there any example questions that utilize one vs the other to show.
QUID forSea Level TAC DUACS Ref CMEMSProducts SEALEVEL_*_PHY_L3/4_NRT/REP_OBSERVATIONS_008_0* Date SL QUID 008 032 062 10 Sept Issue 42. Since work argument above P(V2 V1) = RT is simple and holds for all gases, This suggests KE > (3/2)RT for diatomics, This would make Cp/Cv < 167 Equipartition Theorem This is a very general law which. (always true) 2 Monoatomic ideal gases If U is the internal energy, C_p and C_v are molar heat capacities at constant volume and constant pressure, respectively, U=3/2 nRT C_p = 5/2R;\qquad C_v=3/2R;\qquad \gamma=C_p/C_v=5/3;.
For diatomics and polyatomics find Cp/Cv < 167!. ACROBiosystems is an internationally recognized manufacturer of recombinant proteins committed to supporting antibody development. (How much volume does 1 mole of gas occupy?) nRT (1000 mol)(0006 L atm/mol K)(K).
Explained below 1 Ideal Gas EoS \quad PV=nRT;. K tot trans = N 1 2 m ¯ v 2 = 3 2 NkT = 3 2 nRT (347) If the gas only has translational kinetic energy, this is the internal energy of the gas – this is true for ideal monoatomic gases (one atom per molecule) This tells us that the internal energy of an ideal gas depends only on the temperature The root mean square (rms) speed is the square root of the average of the squares of the.

Dr Molecular Hazlett Mandeville High School Ppt Download
Web Pa Msu Edu Courses 11fall Phy215 Handouts Kinetic Theory Nq Pdf

How To Calculate The Average Translational Kinetic Energy Of Molecules Using Boltzmann S Constant Youtube

Temperature And Kinetic Theory Ppt Video Online Download

Specific Heat Of Gases N Moles Of An Ideal Diatomic Gas Are In A Cylinder At Temperature T Suppose On Supplying Heat To The Gas Its Temperature Remain Constant But N Moles

Thermal Properties Of Matter Ppt Video Online Download

Neet Exam How Is It 3 2 Nrt Chemistry Meritnation Com
Http Markwilde Com Teaching 19 Fall Phys2112 Exam 1 Exam1 Solutions F19 Pdf

What Is Average Kinetic Energy Of 1 Mole Of So2 At 300k Brainly In

Proof U 3 2pv Or U 3 2nrt In Bangla Youtube

16 A Closed Cylindrical Vessel Contains N Moles Of An Ideal Diatomic Gas At A Temperature T On Supplying Heat Temperature Remains Same But In Moles Get Dissociated Into Atoms The Heat

What Is Average Kinetic Energy Of 1 Mole Of So At 300 K 1 4578 J Mol 2 3134 J Mol 4 4173 J Mol 3 Brainly In

Ppt Heat Powerpoint Presentation Free Download Id

Solved P Atm 3 A Monatomic Gas N 0 Moles Undergo Chegg Com
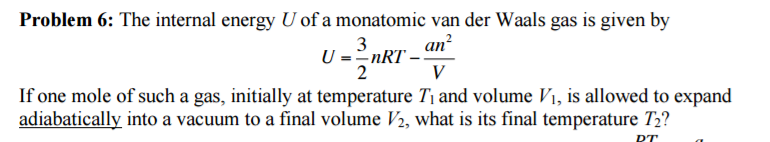
Solved The Internal Energy U Of A Monatomic Van Der Waals Chegg Com

Consider An Equimolar Mixture Of Monoatomic Gas And Diatomic Gas The Heat Required To Increase The
What Is The Average Velocity Of Atoms In 2 00 Mol Of Neon A Monatomic Gas At 308 K Use The Brainly Com
1st Law

Proof U 3 2 Pv Or U 3 2 Nrt Courses Com

Lecture 26purdue University Physics 21 Lecture 26 Thermodynamics I Physics Ppt Download

Derive The Equation U 3 2 Nrt In Thermodynamics Edurev Class 11 Question
Www Kpu Ca Sites Default Files Downloads Pset08 Pdf

For An Ideal Monatomic Gas U 3 2 Nrt So Q U0394 U 32 Nr U0394t 35 E Ifw Is The Work Course Hero

Ib Physics Unit 3 Modelling A Gas Notes

Proof U 3 2 Pv Or U 3 2 Nrt Free Download Borrow And Streaming Internet Archive
2

Univariate Factors Related To Cessation At 38 Weeks N 67 Download Table

Solved Useful Information For An Ideal Gas The Internal Chegg Com
Q Tbn And9gcqlkxpndfttx Gnn4cu8l9rgp9bomqsbcjd2suov0mxxgsmxbin Usqp Cau

Solved What Is The Internal Energy Of 1 0 Mole Of A Monat Chegg Com
2

Boddeker S Phy122 Ch 18 Lecture
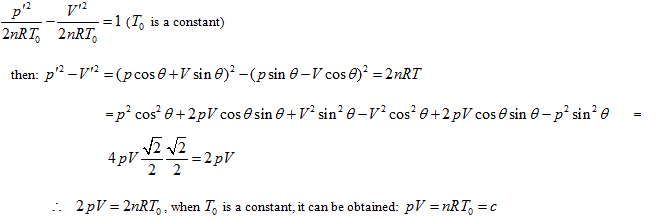
State And Equal Intrinsic Energy Curved Surfaces Of Ideal Gas
Q Tbn And9gcqydv7umvmzpsssheiiluymavmruyefuqldgudklenw5mdln8tk Usqp Cau

Prove That The K E Per Unit Volume Of An Ideal Gas Is 3 2 P

Solved Find Partialdifferential V Partialdifferential P 2 Chegg Com

02 Total Kinetic Energy Of One Mole Of An Ideal Gas At 127 C Is A 8 31 Kj B 4 99 Kj Correct Answer 3 Kj D5 68 Kj Your Answer

Internal Energy Ideal Gas Monatomic Diatomic Gas Nuclear Power Net

Ppt Welcome Back To Physics 211 Powerpoint Presentation Free Download Id
Http Www Physics Sfsu Edu Wman Phy111hw Lecture notes Chapter18 Pdf

Solved Hi Please Provide The Correct Answer Asap This I Chegg Com

Chemistry 2374a Lecture Notes Fall 17 Lecture 8 Gas Constant Sign Convention Adiabatic Process

Thermodynamics Notes Pls Subscribe My Physics In Short And Easy Way Facebook

Derivation Kinetic Energy 3 2 Nrt Youtube

Physicslab State Variables

Solved Problem 3 Adiabatic Expansion Of An Ideal Gas Th Chegg Com
2
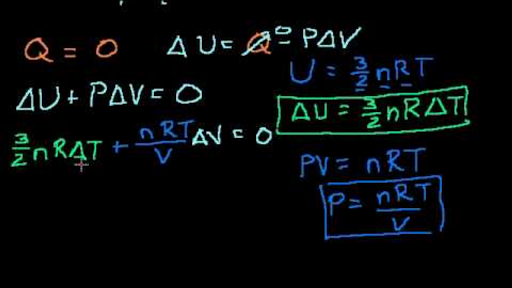
Thermodynamics Physics Library Science Khan Academy
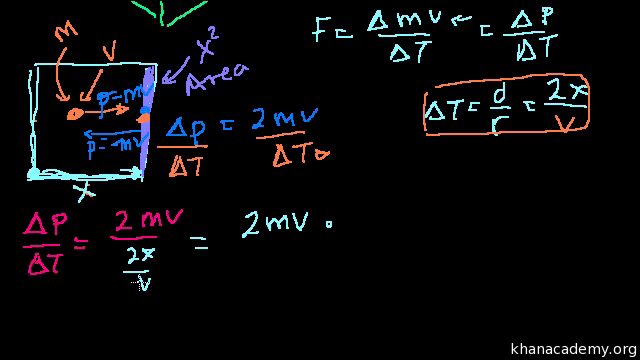
Proof U 3 2 Pv Or U 3 2 Nrt Video Khan Academy

More Thermodynamics Specific Heats Of A Gas Equipartition Of Energy Ppt Download
Http Www Santarosa Edu Lwillia2 41 41ch18 S15 Pdf

Derivation Kinetic Energy 3 2 Nrt Youtube

Solved 1 Liter Of An Ideal Gas Is Allowed To Expand At Co Chegg Com
Q Tbn And9gcrzw0b2pjyyjbfa1rv6n1bss6pypqipf71xguwgugkg8uggm0fr Usqp Cau

Proof U 3 2 Pv Or U 3 2 Nrt Video Khan Academy

Ppt Physics 101 Chapter 15 Thermodynamics Part I Powerpoint Presentation Id

Thermodynamic Part 2

1st Law
Http Www Uvm Edu Jgoldber Courses Chem35 Newfiles Chem35 00 Ch10 Gases Pdf
2

Work Done By Isothermic Process Video Khan Academy

Solved N Moles Of An Ideal Gas With Internal Energy U Chegg Com

The Equation Of State For A Gas Is Given By Pv Nrt Alpha V Where N Is The Number Of Moles And Alpha Is A Positive Constant The Initial

Lecture27
Q Tbn And9gct3katics6yargnn2kudt2paal2vu Mqmhwe4kerqcy5ll2qcrr Usqp Cau

Ppt Lecture 2 Enthalpy Powerpoint Presentation Free Download Id
What Would Be The Average Kinetic Energy Of A Hydrogen Molecule At 300k Quora

Physics 101 Lecture 25 Thermodynamics Ppt Download

Solved Density M V Pressure P F A Relation Between Chegg Com
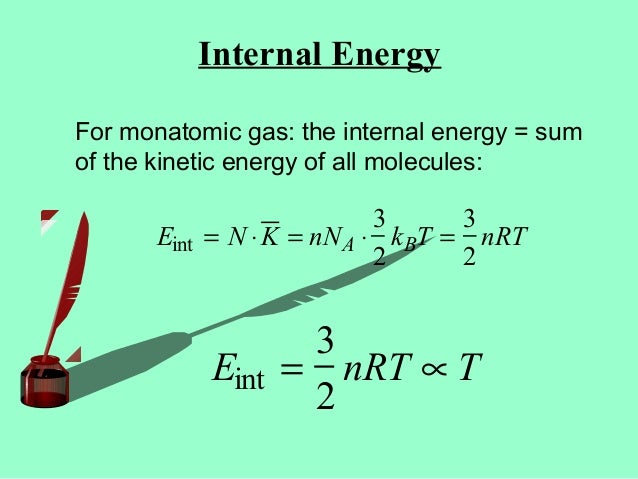
Kinetic Theory Of Gases

Internal Energy Ideal Gas Monatomic Diatomic Gas Nuclear Power Net

Thermos And Then The Thermos Were Shook So As To Break The Seal Within The Cold Course Hero

Solved 12 Ideal Gas Law Pv Nrt What Is R Value A 8 Chegg Com

32 A Sample Contains N Moles Of A Diatomic Gas At Temperature T Molecules Of Gas

General Physics L02 Paths Ppt Energy Transfers Ppt Download

Boddeker S Phy122 Ch 18 Lecture

Chem 123 Study Guide For Physical Chem State Variables Intensive The Ratio Of Extensive Variables Is Intensive Density Ii Temperature Pressure Iv Molar Mass Studocu

Proof U 3 2 Pv Or U 3 2 Nrt Video Khan Academy

Ppt Chapter 14 Heat Powerpoint Presentation Free Download Id

Neet Exam How Is It 3 2 Nrt Chemistry Meritnation Com
2

Proof U 3 2 Pv Or U 3 2 Nrt Thermodynamics Physics Khan Academy Youtube
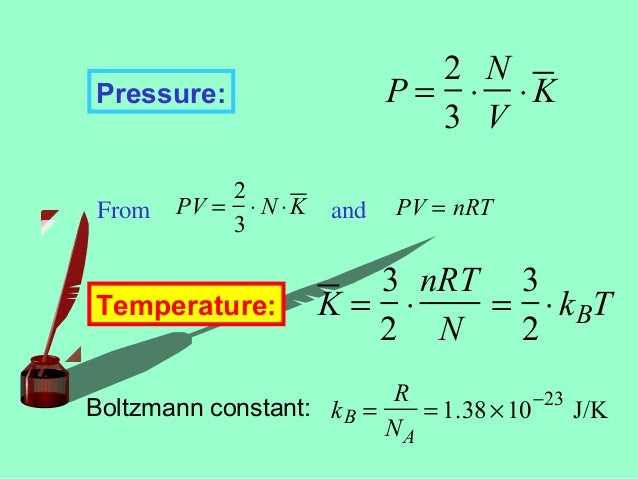
Kinetic Theory Of Gases

Mean Ittq Score By Type Of Facility Download Table

0 Since U 3 2 N R T For An Ideal Gas So Q W 1600 J A Gas Course Hero

Find The Kinetic Energy In Kilo Joules Of 2 Moles Of An Ideal Gas At 00k Brainly In
2

Solved 18 In Monoatomic Gas The Internal Energy U Is Chegg Com

A Closed Cylindrical Vessel Contains N Moles Of An Ideal Diatomic Gas At A Temperature T On Supplying Heat Temperature Remains Same But N Moles Get Dissociated Into Atoms The Heat Supplied

Solved 2 16 Moles Of An Ideal Gas Are Held At A Pressure Chegg Com
2

Consider An Equimolar Mixture Of Monoatomic Gas And Diatomic Gas The Heat Required To Increase The
2

44 What Is The Ratio Of The Average Molecular Energy Of Ufg To That Of H Both At 300 K 1 1 1 2 349 2 3 2 349 4 None Of These

Solved Part 2 Gas Calculations Ke 3 2nrt R 0 006 L A Chegg Com
Http Www Santarosa Edu Lwillia2 Ch15 Pdf

Internal Energy Thermodynamics For Iit Jee Unacademy
3 Referring To Problem 4 Of Problem Set 4 A Ca Chegg Com



