32nkbt
2
Www Physics Nus Edu Sg Phywjs Pc52 X1 Pdf
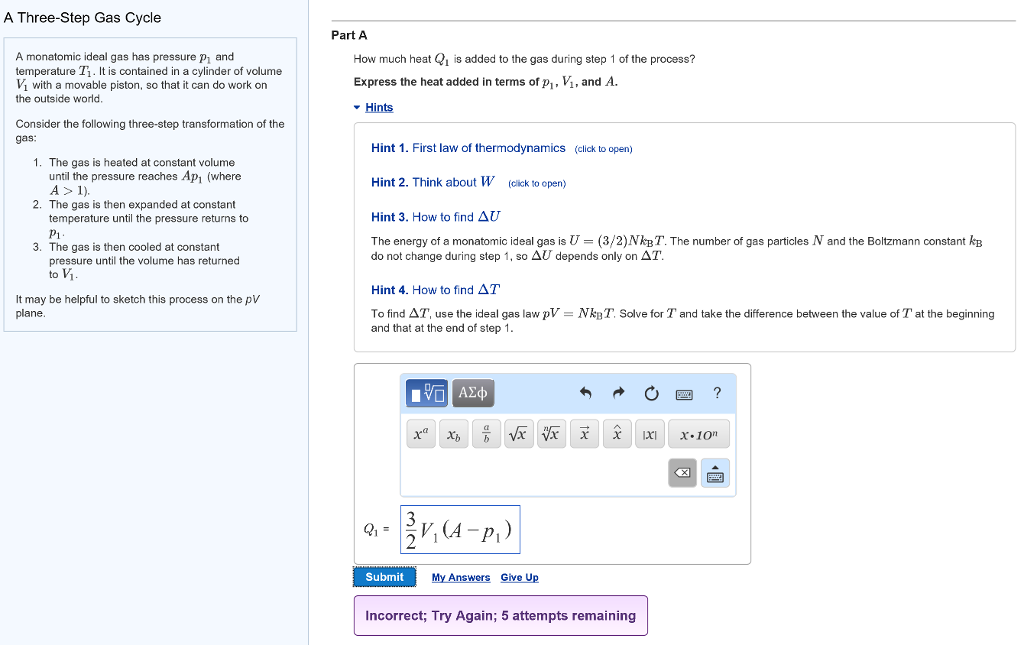
Solved A Three Step Gas Cycle Part A A Monatomic Ideal Ga Chegg Com
2

F11 Physics 231 Lectures 29and Crap

Kinetic Molecular Theory Of Gases Flashcards Quizlet
CiteSeerX Document Details (Isaac Councill, Lee Giles, Pradeep Teregowda) The heat theorem (ie the second law of thermodynamics or the existence of entropy) is a manifestation of a general property of hamiltonian mechanics and of the ergodic Hypothesis In nonequilibrium thermodynamics of stationary states the chaotic hypothesis plays a similar role it allows a unique determination of.

32nkbt. Spero qualcuno ne sia capace. Comme il y a n fois trois composantes quadratiques, on a E_tot = 3/2nKbT Ce que je veux dire est que KbT est juste un facteur qui nous renseigne sur l'énergie thermique dont la température est le reflet Pour avoir la valeur exacte de l'énergie, les calculs sont plus compliqués. A tank of volume 0413 m3 contains 279 mol of helium gas at 5C Assume that the helium behaves like as an ideal gas The universal gas constant is 1451 J/Kmol, and Boltzmann's constant is x10^23 J/K Find the total thermal energy of the system Answer in units of J Would.
3 2NkBT a XNi1 1 2mmi u2 8 where Ta mea Recommended Fourier law Conservation of energy The geotherm Heat Transfer I The main observations and principles of heat conduction Important This chapter follows. T= (2x568)/ (975x10^22 x 13x10^23 x3) =298K The value given in the solution is 2K. Dinámica a T constante La temperatura del sistema se relaciona con el promedio temporal de la energía cinética <K>NVT = 3/2NkBT 1)Escalamiento de velocidades Se multiplican las velocidades por un cierto factor (λ) λ = (T o / T (t )) T0 = temperatura de referencia T(t) = temperatura instantánea ⎡ mi( 2.
The lattice kinetic energy (KE) is canceled out when calculating the energy difference because KE equals 3/2NkBT (N = number of atoms in the system) For oxides, the vibrationalentropic contribution is often small as compared with the defect formation energy 62, 63 For instance, it has been demonstrated that the vibrational contribution to. My thinking was this PAd = some tiny work done by n moles of the gas, d is a tiny distance moved v = Ad = tiny volume expanded or contracted N/n = ratio between total moles. Ec=3/2NKbT M=3NKbT/V^2 C'est une masse aussi moyenne Oui Sinon, vu les hypothèses (molécules identiques), il n'est pas utile de calculer une masse moyenne Si vous lisez le wiki, vous pouvez voir que la vitesse moyenne dépend de la distribution des vitesses des particules (dans une triple intégrale) Avec votre modèle, vous supposez une.
The rms speed of the atoms in a 2g sample of helium gas is 7m/s What is the thermal energy of the gas?. هستش که kB همون ثابت بولتزمان هست و T دمای ِ سیستم برای ِ سیستم ِ تک اتمیی شامل ِ N اتم، 3N درجه ی ِ آزادی داریم که قضیه ی ِ همپاری می گه انرژی ِ درونی باید 3/2NkBT باید باشه که این رو همه دیدید!. DINAMICA MOLECULAR Mtodos de simulacinPermitir el estudio de propiedades de sistemas complejosGeneracin de conjunto de configuraciones distintas para un mismo sistemaPrediccin del comportamiento temporal de un sistema (MD) Dos mtodosDinmica molecular promedio temporalMonte Carlo promedio de ensamblado Propiedad A dependiente de las posiciones y momentos de todas las partculas.
• The total translational kinetic energy of N molecules of gas is simply N times the average energy per molecule Etrans = N(1/2mv2average) = 3/2NkBT = 3/2nNA(R/NA)T = 3/2nRT • As kB = R/NA and n = N/NA for the number of moles of gas. PK nNHoa«, mimetypeapplication/epubzipPK Œh}H¥N˘‡H u˜% EPUB/Content/xhtmlì}Û’ ǵå³ù 5í 2 @WÞê"“ AC’í㦬±l 8q¢ ¨n”‰›Q. บทที่ 16 ความร้อน สมบัติของก๊าซและทฤษฎีจลน์ ความร้อนและพลังงาน อุณหภูมิ คือ “ระดับความร้อน” เราวัดอุณหภูมิด้วยเครื่องม.
PK nNHoa«, mimetypeapplication/epubzipPK Œh}H¥N˘‡H u˜% EPUB/Content/xhtmlì}Û’ ǵå³ù 5í 2 @WÞê"“ AC’í㦬±l 8q¢ ¨n”‰›Q. In più, tutti dicono che essa dipende solo dalla temperatura, ma nessuno spiega il perché!. Get in touch with Martina Nese (@MartinaNese) — 1237 answers, 2693 likes Ask anything you want to learn about Martina Nese by getting answers on ASKfm.
บทที่ 16 ความร้อน สมบัติของก๊าซและทฤษฎีจลน์ ความร้อนและ. 第二章 §21 §22 §23 §24 §25 §26 §27 能带论(Band Theory) 能带论的基本假定与Bloch定理 KronigPenney模型 近自由电子近似 紧. Chapter the first law of thermodynamics outline what is the 1 st law of thermodynamics internal energy (e) heat.
For thermal energy density (u th = 3/2P th = 3/2nkBT ), kinetic temperature is assumed to be ∼ Tex, and typical n(H2) ∼ 10 6 cm −3 in the this PDR region this results in an energy density of. 交换后, 副本动 量按 pi ,new=(Tnew / Told )05 pi ,old 进行调整, Told 和 Tnew 分别 是交换前和交换后的温度 这确保平均动能仍然等于 3/2NkBT 值得注意的是, 副本交换是 “非物理” 过程, 只 能作为有效采样的方法, 并不能从中得到真实的动力学 结论. Yahoo Answers I know that Eth= 3/2NkbT=3/2nRt But, where exactly does that helium.
物理について質問です。 回答お願いたします。 質問1 1気圧のもとで、℃の水1kgを100℃の水蒸気にするために必要な熱量は. Average Energy Integral Boltzmann Distribution The average energy integral for the distribution of energy among a collection of particles according to the Boltzmann distribution is This integral may be evaluated using integration by parts This technique is particularly appropriate for removing a linear term multiplying an exponential. DINAMICA MOLECULAR Mtodos de simulacinPermitir el estudio de propiedades de sistemas complejosGeneracin de conjunto de configuraciones distintas para un mismo sistemaPrediccin del comportamiento temporal de un sistema (MD) Dos mtodosDinmica molecular promedio temporalMonte Carlo promedio de ensamblado Propiedad A dependiente de las posiciones y momentos de todas las partculas.
M46A Introduction to Statistical Mechanics { Wei Cai { Stanford University { Win 11 Handout 9 NPT and Grand Canonical Ensembles January 26, 11. A tank of volume 0413 m3 contains 279 mol of helium gas at 5C Assume that the helium behaves like as an ideal gas The universal gas constant is 1451 J/Kmol, and Boltzmann's constant is x10^23 J/K Find the total thermal energy of the system Answer in units of J Would. CHAPTER 7 CANONICAL AND GRAND CANONICAL ENSEMBLE52 or A = ÈHÍT A ˆA ˆT B V = U ≠TS (711) Therefore once the partition function is obtained it is a straightforward.
エネルギー等分配の法則 (読み)エネルギーとうぶんぱいのほうそく (英語表記)equipartition law of energy. Yahoo Answers I know that Eth= 3/2NkbT=3/2nRt But, where exactly does that helium. Cada subsistema é um gás ideal de N part́ıculas, à mesma temperatura T , com energia E = 3/2NKBT (a) Calcule a entropia total Si de cada gás (i = {1, 2});.
Perchè gli urti sono perfettamente elastici Quindi l'agitazione delle particelle, che si traduce in energia interna, dipende solo da quanto calore hanno a disposizione e quindi dalla temperatura. For thermal energy density (u th = 3/2P th = 3/2nkBT ), kinetic temperature is assumed to be ∼ Tex, and typical n(H2) ∼ 10 6 cm −3 in the this PDR region this results in an energy density of. I rearranged u=3/2NkbT, to get T= (2U)/ (3NKb) Then i changed from mass to molecules, by taking N= (m/M)x (avogardos)= (646/399)x602x10^23=975x10^22 I plotted in the values;.
Start studying Physics Equation Learn vocabulary, terms, and more with flashcards, games, and other study tools. Eint = 3/2 NkT = 3/2 nRT where n is the number of moles Each direction (x, y, and z) contributes (1/2)nRT to the internal energy This is where the equipartition of energy idea comes in – any other contribution to the energy must also contribute (1/2)nRT. Cada subsistema é um gás ideal de N part́ıculas, à mesma temperatura T , com energia E = 3/2NKBT (a) Calcule a entropia total Si de cada gás (i = {1, 2});.
F denotes the degrees of freedom For a monatomic molecule, Eint = 3/2NkBT 95 Adiabatic Processes 951 Definition A process during which no heat leaves or enters the system (Q = 0) Two ways of doing this include o Insulating the walls of the container o Performing the process really quickly From the first law of thermodynamics (∆Eint = Q. 物理について質問です。 回答お願いたします。 質問1 1気圧のもとで、℃の水1kgを100℃の水蒸気にするために必要な熱量は. The rms speed of the atoms in a 2g sample of helium gas is 7m/s What is the thermal energy of the gas?.
The KE=(1/2)mv2 video link is https//wwwyoutubecom/watch?v=7jqnKbeX5E This video is a very quick, all math, derivation of KE=(3/2)nRT=(3/2)PVThis is. บทที่ 16 ความร้อน สมบัติของก๊าซและทฤษฎีจลน์ ความร้อนและ. (3/2)nRT is the translational kinetic energy, and since almost all atoms are in the ground electronic state at low temperature, it is a good expression for internal energy as long as the temperature is low enough that essentially all atoms are in the electronic ground state.
3 2NkBT a XNi1 1 2mmi u2 8 where Ta mea Recommended Fourier law Conservation of energy The geotherm Heat Transfer I The main observations and principles of heat conduction Important This chapter follows. (b) Se os dois gases são o mesmo gás, a entropia S do sistema composto deve ser igual a entropia do sistema composto sem a partição (um gás ideal de 2N part́ıculas à temper. F denotes the degrees of freedom For a monatomic molecule, Eint = 3/2NkBT 95 Adiabatic Processes 951 Definition A process during which no heat leaves or enters the system (Q = 0) Two ways of doing this include o Insulating the walls of the container o Performing the process really quickly From the first law of thermodynamics (∆Eint = Q.
(b) Se os dois gases são o mesmo gás, a entropia S do sistema composto deve ser igual a entropia do sistema composto sem a partição (um gás ideal de 2N part́ıculas à temper. I had always thought the total kinetic energy in a system is PV = nRT, but today at school I saw someone say it was 3/2 nRT or 3/2 PV Can anyone prove this and explain why PV does not yield the kinetic energy of a system?. Under changes b dα of the control parameters the thermodynamic quantities change so that the variation of the average energy and the variation of the volume are dU and dV and are related to the time averages of the kinetic energy 〈K 〉 def = 〈 N m˙x i=1 2 def i 2 〉 = 3 2NkBT and of p def = 〈−∂V U〉, with U being the total.
บทที่ 16 ความร้อน สมบัติของก๊าซและทฤษฎีจลน์ ความร้อนและพลังงาน อุณหภูมิ คือ “ระดับความร้อน” เราวัดอุณหภูมิด้วยเครื่องม. My thinking was this PAd = some tiny work done by n moles of the gas, d is a tiny distance moved v = Ad = tiny volume expanded or contracted N/n = ratio between total moles. Thermodynamics and Statistical Physics Solutions, Chapter 2 216 The number of ways to pick 500 heads and 500 tails is the number of ways to pick 500 heads.
The two equations agree when the average translational kinetic energy of the molecules is K av = (3/2)kT Here we have a fundamental connection between temperature and the average translational kinetic energy of the atoms they are directly proportional to one another Temperature is a measure of the average kinetic energy of the atoms. U = 3/2NKBT = 3/2nRT 4 อุณหพลศาสตร์ (Thermodynamics) 41 กฎข้อที่ศูนย์ของอุณหพลศาสตร์ (Zeroth law of thermodaynamics) 411 ระบบที่มีอุฌหภูมิสูงกว่า จะส่งผ่านความร้อนไป. There are three independent directions in which a gas particle can move (three independent components of velocity), so the total kinetic energy is 3 × k B T / 2.
Thermal Energy Approximating U by U ~ 3/2nkBT ~ mRT/m U/W ~ mRT/m (Gm2/R)1 = 3*103 (m/105Msun)1 (R/25pc) (T/15K) > Clouds cannot be supported by thermal pressure alone!. 열역학에 이어서이다 잘해두면 화학도 잡고 물리도 잡고 딱히 어려운 것은 없음 앞서 말했듯이 단열일때 주의 할것 단열일 때 열의 출입이 없다 그 상황에서 부피가 작아지거나 커진다면 내부의 온도는 올라. Ec=3/2NKbT M=3NKbT/V^2 C'est une masse aussi moyenne Oui Sinon, vu les hypothèses (molécules identiques), il n'est pas utile de calculer une masse moyenne Si vous lisez le wiki, vous pouvez voir que la vitesse moyenne dépend de la distribution des vitesses des particules (dans une triple intégrale) Avec votre modèle, vous supposez une.
I had always thought the total kinetic energy in a system is PV = nRT, but today at school I saw someone say it was 3/2 nRT or 3/2 PV Can anyone prove this and explain why PV does not yield the kinetic energy of a system?. U = 3/2NKBT = 3/2nRT 4 อุณหพลศาสตร์ (Thermodynamics) 41 กฎข้อที่ศูนย์ของอุณหพลศาสตร์ (Zeroth law of thermodaynamics) 411 ระบบที่มีอุฌหภูมิสูงกว่า จะส่งผ่านความร้อนไป. We study supernovadriven galactic outflows as a mechanism for injecting turbulence in the intergalactic medium (IGM) far from galaxies To this aim, we follow the evolution of a 1013M⊙ galaxy along its merger tree, with carefully calibrated prescriptions for star formation and wind efficiencies.
I've already told you multiple times that big, uppercase U is the internal energy of a system And it's really everything thrown in there It's the kinetic energy of the molecules. STATO E TRASFORMAZIONE DEI GAS Per studiare un gas possiamo pensare di confinarne una certa quantità all’interno di un cilindro a tenuta stagna dotato di un pistone mobile e di misurarne la massa (kg) il volume (m*3) il volume di un gas coincide con il volume del recipiente pressione (Pa ovvero N/m*2) temperatura (K o °C). Magnetic energy Approximating M by M ~ B2r3/6p (cloud approximated as sphere) M/W ~ B2r3/6p (Gm2/R)1 = 03 (B/mG)2 (R/25pc)4 (m/105Msun)2 > Magnetic force is.
Comme il y a n fois trois composantes quadratiques, on a E_tot = 3/2nKbT Ce que je veux dire est que KbT est juste un facteur qui nous renseigne sur l'énergie thermique dont la température est le reflet Pour avoir la valeur exacte de l'énergie, les calculs sont plus compliqués. 493 where H is the classical Hamiltonian, h is Planck's constant, and the classical partition function Q is Q = hM ∫ exp ( H(q, p)/kT) dq dp This probability density expression, which must integrate to unity, contains the factor of. DINAMICA MOLECULAR Mtodos de simulacinPermitir el estudio de propiedades de sistemas complejosGeneracin de conjunto de configuraciones distintas para un mismo sistemaPrediccin del comportamiento temporal de un sistema (MD) Dos mtodosDinmica molecular promedio temporalMonte Carlo promedio de ensamblado Propiedad A dependiente de las posiciones y momentos de todas las partculas.
Facendo delle ricerche su internet ho trovato diverse definizioni di energia interna, tra cui DU=3/2nKBT (D= DELTA) e, molto più semplice, DU=QL sono entrambe corrette?.
Web Pa Msu Edu People Duxbury Courses Phy1 Solutions2 12 Pdf

Solved For This Question I Tried All Three Checked Eth Chegg Com

Physics I Gases Flashcards Quizlet
Q Tbn And9gcqk Djjyvp6 Lgbrqsu Ipdpd Pdbve Qityfklip4ied L8jj9 Usqp Cau
Ib Physics Unit 3 Modelling A Gas Notes

Exam 3 Review Byu Department Of Physics And Astronomy Powerpoint Presentation Free Online Download Ppt Ojm70j

Day 4 Kinetic Theory And Pv Diagrams
2
Www Physics Byu Edu Faculty Colton Docs Phy105 Fall14 Exam 3 Review Pdf

Thermal Physics Thermodynamics Relates Heat Work With Empirical Observed Not Derived Properties Of Materials E G Ideal Gas Pv Nrt 2 Statistical Ppt Download
Http Pages Physics Cornell Edu Sethna Teaching 218 F02 Hw Homeworks02 Pdf
Physics Purdue Edu Webapps Index Php Course Document Index Phys515 1905 4

Solution An Ideal Gas Is Described By Pv Physics
Azo Polymers For Holographic Recording Photo Assisted Holography And Surface Relief Gratings

Ch18 The Micro Macro Connection Ppt Download

New Microsoft Word Document Pdf 1 We Know The Equation Is Vrms Sqrt 3kb T M The Kb Is A Constant T Is Room Temperature We Assume That The Volume Of Course Hero
Www Sctimst Ac In Academic and research Academic Previous year questions Resources Entrance examination for phd physical sciences stream Pdf
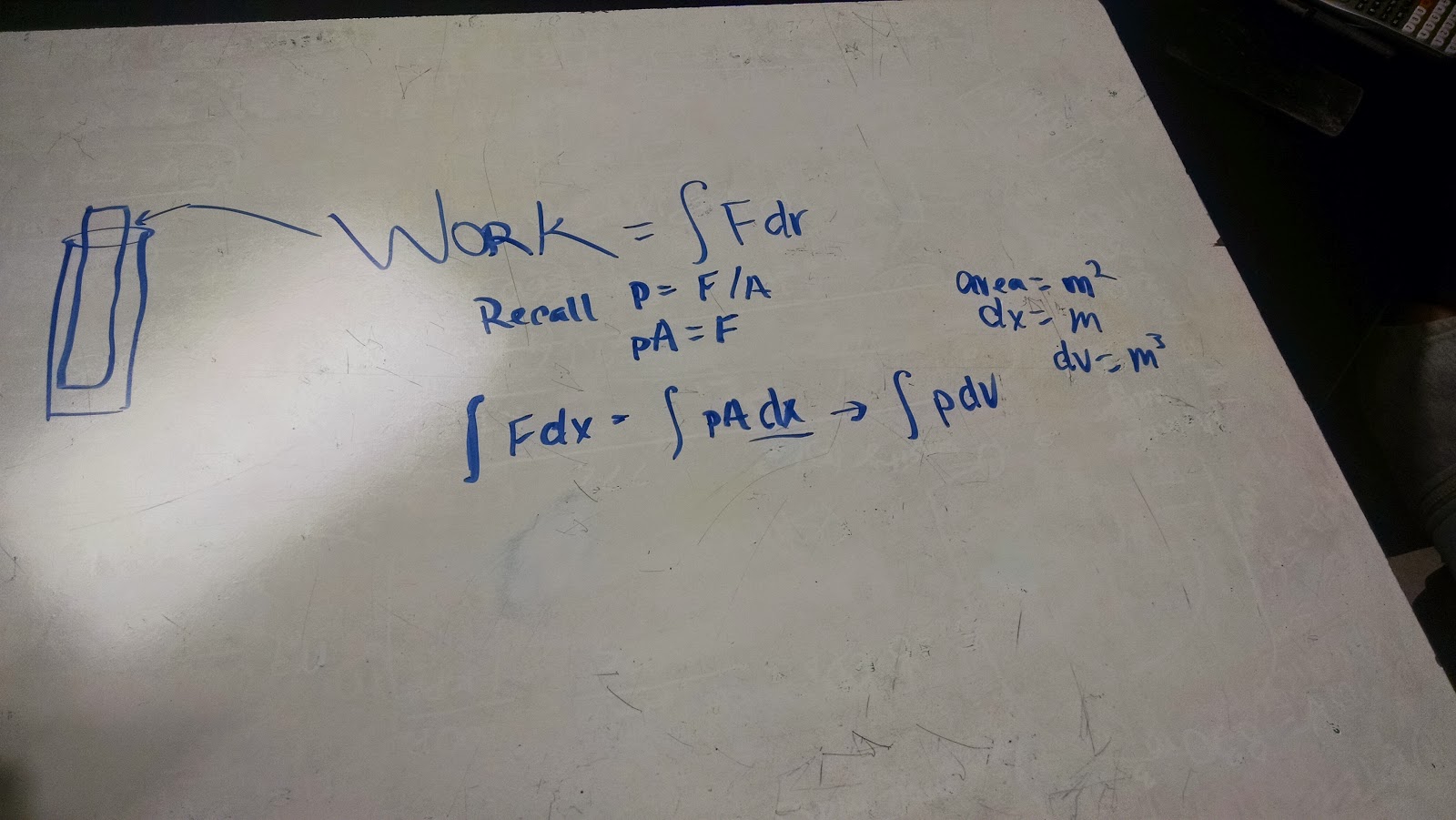
Physics 4b Djharry 9 9 14 Work And The First Law Of Thermodynamics

Ch18 The Micro Macro Connection Ppt Download
2
Maxwell Kinetic Energy Distribution Physics Forums
Http Physics Oregonstate Edu Minote Courses Ph315 Lib Exe Fetch Php Media Gasprocesses Pdf

Spring 13

Answered In A Particular Engine A Gas Is Bartleby

Ch18 The Micro Macro Connection Ppt Download
Ib Physics Unit 3 Modelling A Gas Notes
2

Homework Problems

Solved 1 I For An Ideal Monatomic Gas Of N Particles Th Chegg Com
2

122ch18

Thermal Physics Thermal Physics Is The Study Of Temperature And Heat And How They Effect Matter Heat Leads To Change In Internal Energy Which Shows As Ppt Download

Practice 39 Mtb Doc

New Microsoft Word Document Pdf 1 We Know The Equation Is Vrms Sqrt 3kb T M The Kb Is A Constant T Is Room Temperature We Assume That The Volume Of Course Hero
Http Mini Physics Sunysb Edu Marivi Wordpress Wp Content Uploads 13 02 Samplemt1sol Pdf

Proof U 3 2 Pv Or U 3 2 Nrt Video Khan Academy
Www Physics Byu Edu Faculty Colton Docs Phy105 Fall14 Exam 3 Review Pdf
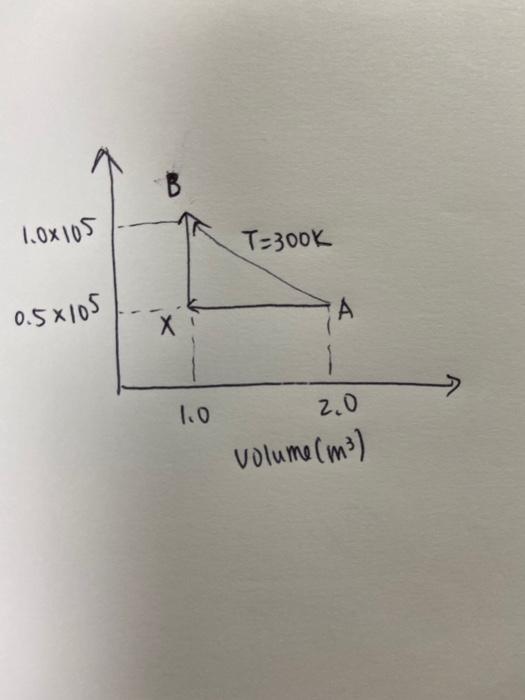
Solved A Monoatomic Ideal Gas Shows Reversible Process Sh Chegg Com

Solved An Ideal Gas With Energy E 3 2 Nk G T Moves Quas Chegg Com
Q Tbn And9gctdxopxkif00upj7tdi Rty6fut5dxboprtzqzognwu Ctx 0sw Usqp Cau

Temperature And Kinetic Theory Ppt Video Online Download

Drude S Classical Model Of Metals A Theory Of Metallic Conductivity Ppt Download
2
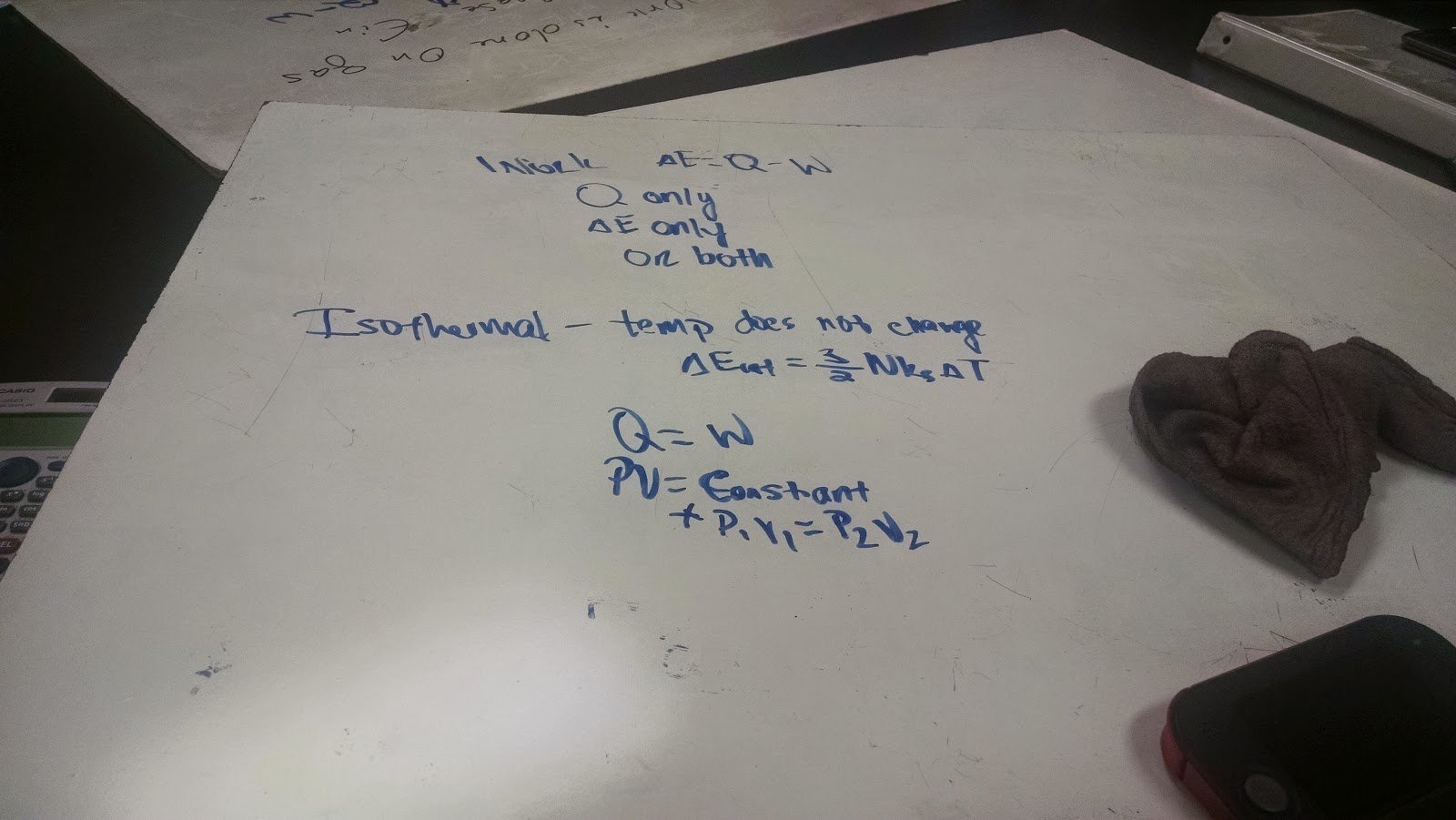
Physics 4b Djharry 9 9 14 Work And The First Law Of Thermodynamics
2
Http People Bss Phy Cam Ac Uk Emt1000 Statphys Handout12 Pdf
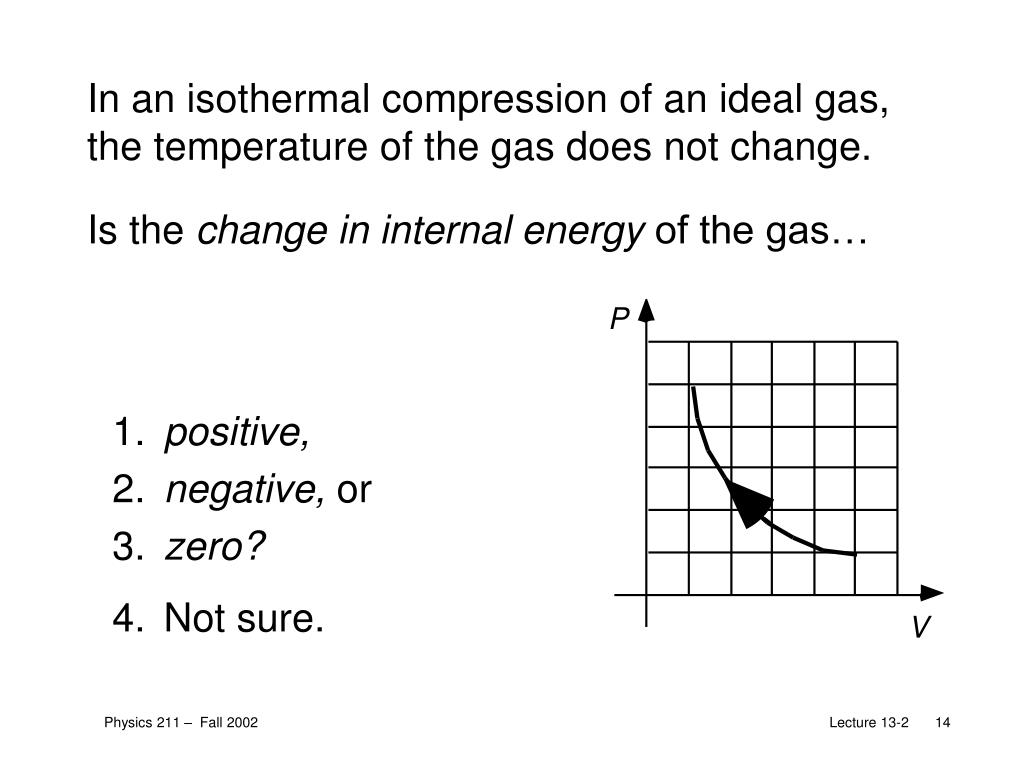
Ppt Welcome Back To Physics 211 Powerpoint Presentation Free Download Id
Http Www Physics Umd Edu Courses Phys404 Einstein Fall14 Phys404midtermf13 Pdf
2
2

Solved 2 Describe Whether The Following Equations Apply Chegg Com

Solved Problem 5 The First Law Of Thermodynamics Can Be Chegg Com
Http Link Aps Org Pdf 10 1103 Physreve 54 2501
2

Solved 2 10 2 Ideal Gas In The Microcanonical And Canonic Chegg Com
2
Q Tbn And9gcshwlqj7jl3gtmc3hjxriqmiakeixjujtbhyuckmj2g0a4chsfk Usqp Cau

Physics 231 Lecture 29 Main Points Of Today S Lecture Temperature Ppt Download
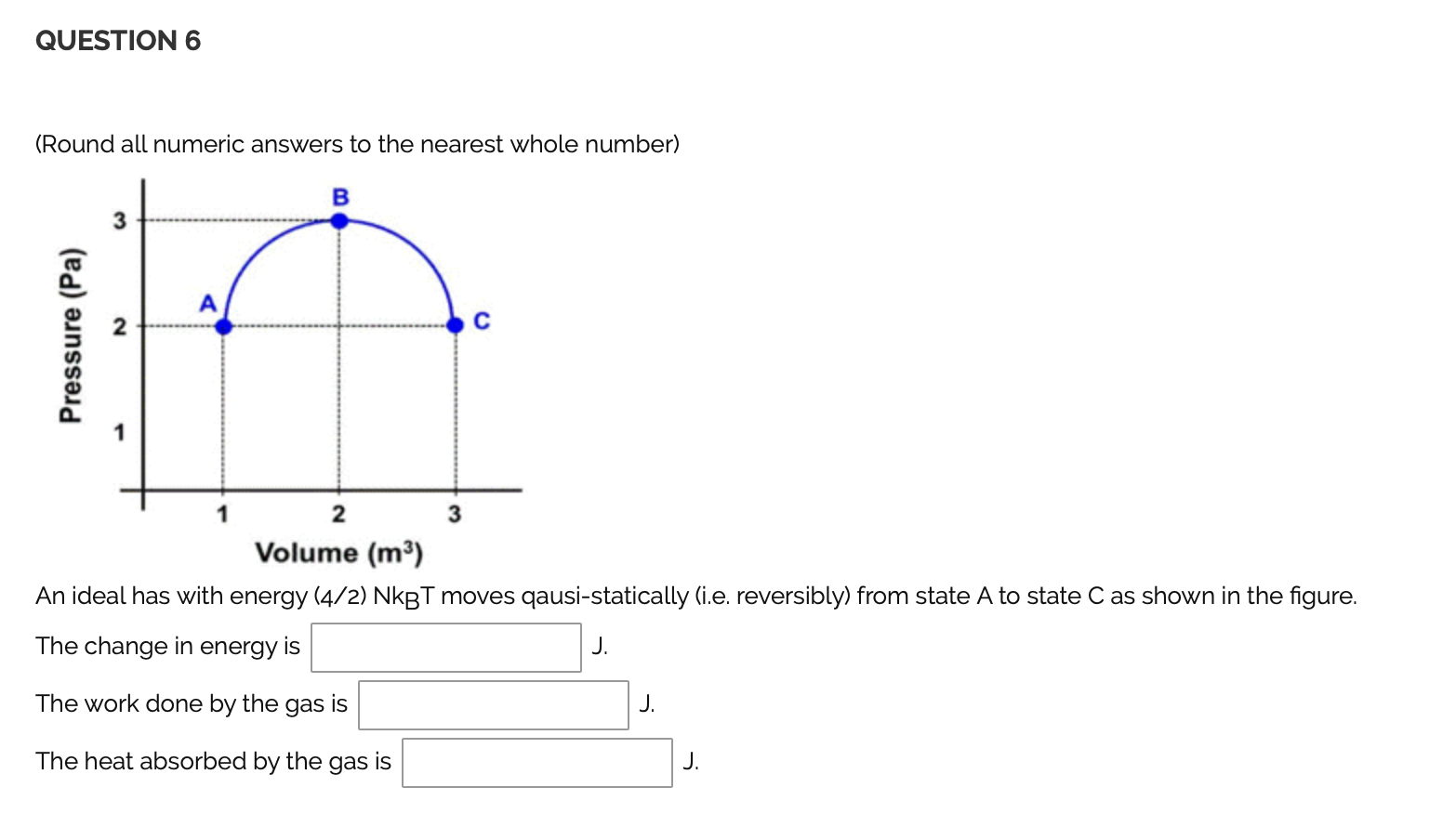
Solved Question 6 Round All Numeric Answers To The Neare Chegg Com

Umdberg Example Degrees Of Freedom

Solved 18 Objective 6 A Total Of 1 X 1024 Molecules Of Chegg Com

Physics I Gases Flashcards Quizlet
Arxiv Org Pdf 1008 0448

New Microsoft Word Document Pdf 1 We Know The Equation Is Vrms Sqrt 3kb T M The Kb Is A Constant T Is Room Temperature We Assume That The Volume Of Course Hero

Ch18 The Micro Macro Connection Ppt Download

Ch18 The Micro Macro Connection Ppt Download
2
2

Temperature And Kinetic Theory Ppt Video Online Download

Physics 4b Mcecardora Work

Physics 1 Flashcards Quizlet

Physics 4b Mcecardora Work
Ocw Aprende Org Courses Physics 8 333 Statistical Mechanics I Statistical Mechanics Of Particles Fall 13 Exams Mit8 333f13 Examreview3sol Pdf
2

Solved Use The Statistical Mechanical Expression For The Chegg Com
Www Sctimst Ac In Academic and research Academic Previous year questions Resources Entrance examination for phd physical sciences stream Pdf

Solved 7 This Problem Involves The Maxwell Boltzmann Spe Chegg Com
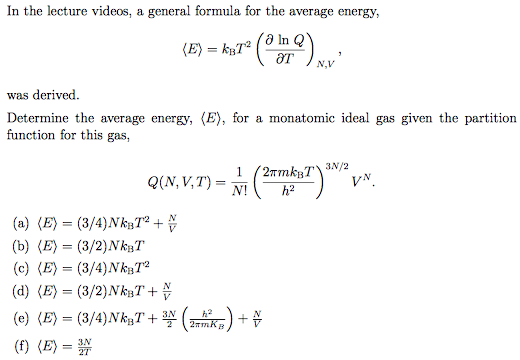
Solved In The Lecture Videos A General Formula For The A Chegg Com
Ib Physics Unit 3 Modelling A Gas Notes
2

Solved 1 I For An Ideal Monatomic Gas Of N Particles Th Chegg Com
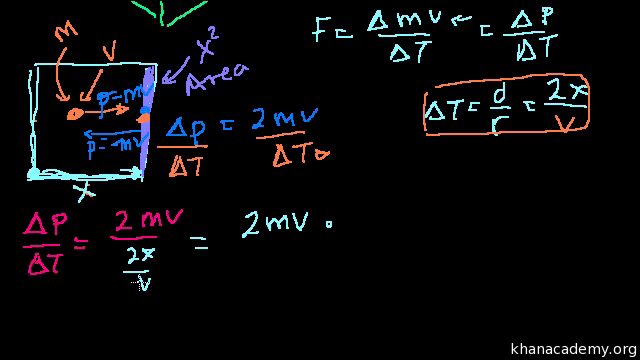
Proof U 3 2 Pv Or U 3 2 Nrt Video Khan Academy
Www Physics Byu Edu Faculty Colton Docs Phy105 Fall14 Exam 3 Review Pdf
Q Tbn And9gcqdffvtmmeiosatbgznaihvcnj4j7qdyaqh0ufsehmz3mbxxgzp Usqp Cau

Umdberg Example Degrees Of Freedom
2
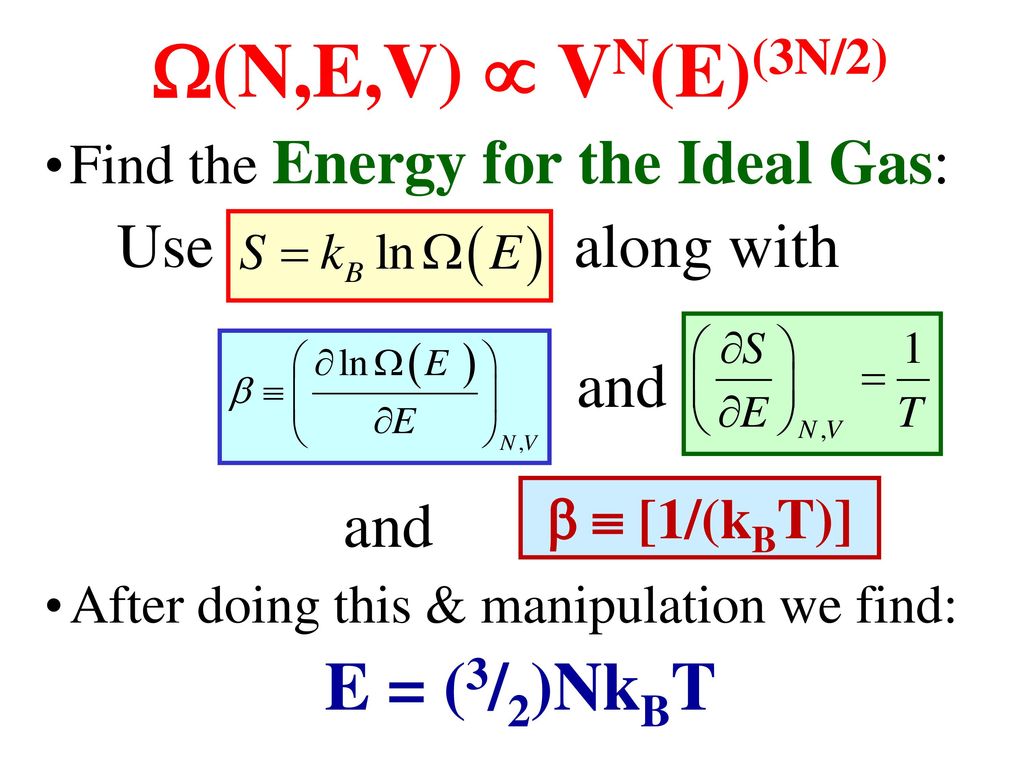
Classical Ideal Gas Ppt Download

Physics 4b Djharry 9 9 14 Work And The First Law Of Thermodynamics
Arxiv Org Pdf Gr Qc
Maxwell Kinetic Energy Distribution Physics Forums

The Segment Cd Is Also An Isothermal Process Answer U B U A U D U C 0 0 Part F Course Hero

Temperature Temperature T Measure Of The Average Kinetic Energy Of The Particles Composing An Object Or System Due To Vibrations Rotations Random Translations Ppt Video Online Download
2

Gases And Heat Chapter Ppt Video Online Download

Wedushare វ ឌ ស រ Source Moeys Gov Kh Correction Kav 3 2kbt K 3 2nkbt 3 2nrt 3 2pv Facebook

Physics I Gases Flashcards Quizlet

Derivation Kinetic Energy 3 2 Nrt Youtube

Proof U 3 2 Pv Or U 3 2 Nrt Video Khan Academy



